Furan is an organic compound with the molecular formula C4H4O. It is a five-membered ring consisting of four carbon atoms and one oxygen atom. It is an important intermediate in the production of many chemicals, including pharmaceuticals. This article will discuss the number of pi electrons in furan, its structure, and its properties.
Overview of Furan
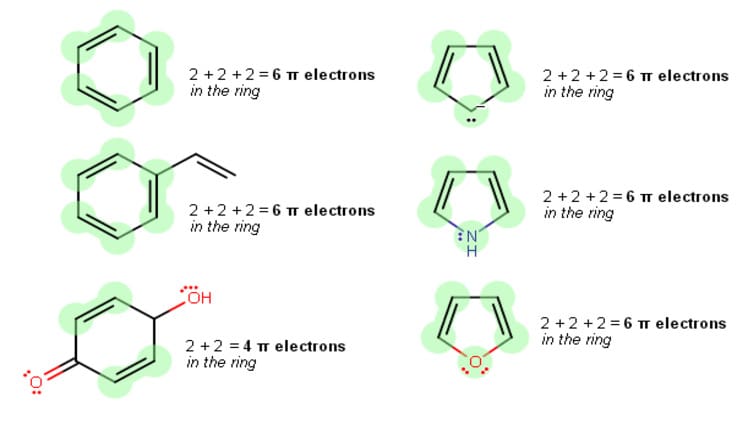
Furan has a planar, cyclic structure with an oxygen atom in the center of the ring. The four carbon atoms are sp2-hybridized, meaning they each have three sp2-hybridized orbitals and one unhybridized p-orbital. The unhybridized p-orbitals are arranged perpendicular to the plane of the ring and form a pi system. This pi system consists of four pi electrons, two of which are delocalized over the entire ring.
Number of Pi Electrons in Furan
Furan has four pi electrons. Two of these electrons are in the form of a single pi bond between the oxygen atom and one of the carbon atoms. The other two electrons are delocalized over the entire ring and are known as “conjugated pi electrons”. These conjugated pi electrons give furan its unique properties, such as its high reactivity and its ability to act as an electron acceptor.
In conclusion, furan is an important intermediate in many chemical processes. It has a planar, cyclic structure with four sp2-hybridized carbon atoms and one oxygen atom in the center. The unhybridized p-orbitals form a pi system with four pi electrons, two of which are delocalized over the entire ring. These conjugated pi electrons give furan its unique properties.