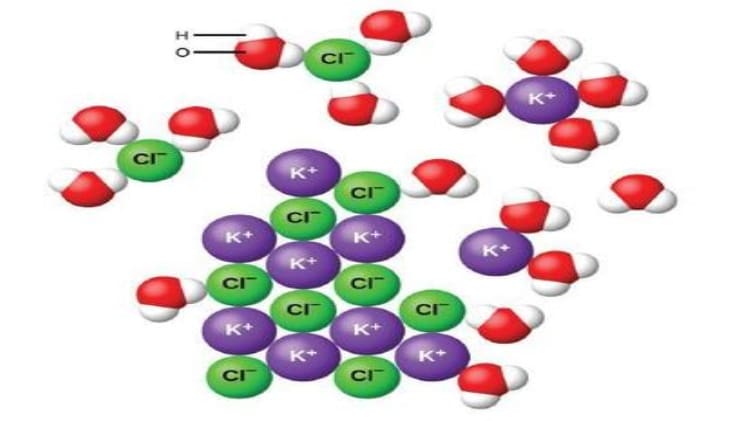
KCl, also known as potassium chloride, is a compound that is commonly used in many industrial and medical applications. It is also found in nature, and is a major part of the salt found in seawater. Potassium chloride is highly soluble in water, which makes it an ideal choice for many applications. In this article, we will discuss why KCl is so soluble in water.
Solubility of KCl
KCl is highly soluble in water, with a solubility of 74.5 g/L at 25°C. This is due to the fact that the ions of potassium and chloride are attracted to the polar water molecules. The ions are small and have a high charge, making them more attracted to the water molecules. This attraction between the ions and the water molecules causes the KCl to dissolve in the water.
Reasons for KCl’s Solubility in Water
- Polar Water Molecules: KCl is highly soluble in water because the ions of potassium and chloride are attracted to the polar water molecules. The ions are small and have a high charge, making them more attracted to the water molecules.
- Electrostatic Attraction: The attraction between the ions and the water molecules causes the KCl to dissolve in the water. This is due to the electrostatic attraction between the positively charged potassium ions and the negatively charged chloride ions.
- Hydrogen Bonding: The hydrogen bonds between the molecules of KCl and the water molecules also contribute to the solubility of KCl in water. The hydrogen bonds help to keep the molecules together, which makes it easier for them to dissolve in the water.
- Low Melting Point: The melting point of KCl is also relatively low, which makes it easier for the molecules to dissolve in the water. This is because the lower temperature makes it easier for the molecules to break apart and dissolve.
In conclusion, KCl is highly soluble in water due to the attraction between the ions and the polar water molecules, the electrostatic attraction between the positively charged potassium ions and the negatively charged chloride ions, the hydrogen bonding between the molecules of KCl and the water molecules, and the low melting point of KCl. This makes KCl an ideal choice for many industrial and medical applications.