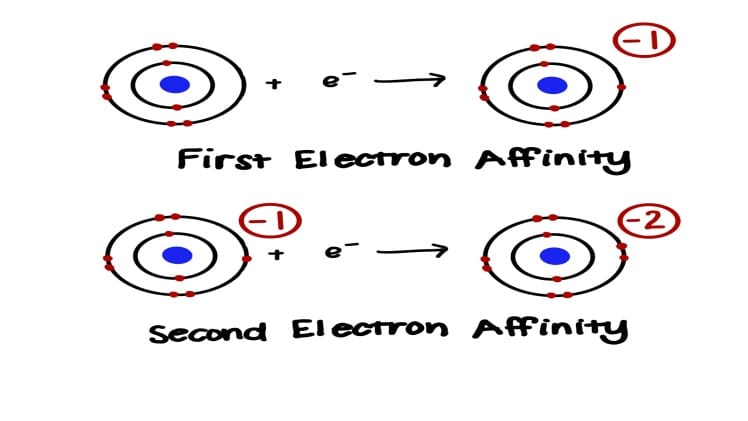
The 2nd electron affinity is a measure of the amount of energy released when an additional electron is added to a neutral atom in the gaseous state. It is a critical parameter in the study of atomic and molecular properties, and understanding why it is positive is important for a wide range of scientific applications.
Understanding 2nd Electron Affinity
The 2nd electron affinity (EA2) is the amount of energy released when an additional electron is added to a neutral atom in the gaseous state. It is usually expressed in units of kJ/mol and is a measure of the strength of the electrostatic force between the additional electron and the atom. Generally, the higher the EA2, the greater the electrostatic force between the electron and the atom.
When an additional electron is added to a neutral atom, the additional electron will be repelled by the positively charged nucleus of the atom. This repulsion will result in the release of energy, which is the 2nd electron affinity.
Reasons for Positive 2nd Electron Affinity
- Electron-electron repulsion: When an additional electron is added to a neutral atom, the existing electrons will repel the new electron due to their negative charges. This repulsion will result in the release of energy, which is the 2nd electron affinity.
- Electron-nucleus attraction: The additional electron will also be attracted to the nucleus of the atom due to its positive charge. This attraction will result in the release of energy, which is the 2nd electron affinity.
- Electron shielding: Electrons in an atom are arranged in shells and each shell has a certain amount of shielding, or protection, from the nucleus. This shielding will result in the release of energy, which is the 2nd electron affinity.
In conclusion, the 2nd electron affinity is positive due to electron-electron repulsion, electron-nucleus attraction, and electron shielding. Understanding why the 2nd electron affinity is positive is important for a wide range of scientific applications.