Electron loss is a common phenomenon in atoms, in which electrons are lost from the outer shell of an atom. The process of electron loss is important in many areas of science, such as chemistry, physics, and materials science. By understanding which elements are likely to lose electrons, we can gain insight into the behavior of atoms and molecules in various environments.
Electron Loss in Elements
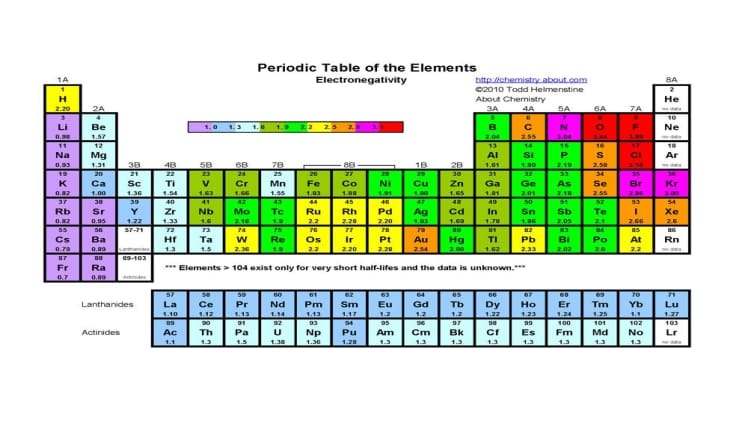
Electron loss is most likely to occur in elements with high electronegativity, or the ability of an atom to attract electrons. Elements with high electronegativity include oxygen, fluorine, chlorine, and nitrogen. These elements are more likely to form bonds with other atoms, leading to the transfer of electrons from one atom to another. When an atom loses electrons, it becomes positively charged and is called a cation.
In addition to elements with high electronegativity, elements with low ionization energies are also likely to lose electrons. Ionization energy is the energy required to remove an electron from an atom, and elements with low ionization energies are easier to remove electrons from. Examples of elements with low ionization energies include lithium, sodium, and calcium.
What Factors Affect It?
The likelihood of electron loss is also affected by the environment in which the atom is located. In gases, electrons are more likely to be lost than in solids. This is due to the fact that in gases, atoms are more likely to move around and collide with each other, leading to electron transfer. In addition, the presence of other atoms or molecules in the environment can also affect electron loss. For example, when an element is in a solution, the presence of other molecules can increase the likelihood of electron transfer.
Finally, the temperature of the environment can also affect electron loss. At higher temperatures, atoms move around more rapidly, increasing the chance of electron transfer. On the other hand, at lower temperatures, electrons are less likely to be lost due to the decreased movement of atoms.
In summary, electron loss is most likely to occur in elements with high electronegativity and low ionization energies. The environment in which the atom is located, as well as the temperature, can also affect the likelihood of electron loss. By understanding which elements are likely to lose electrons, we can gain insight into the behavior of atoms and molecules in various environments.