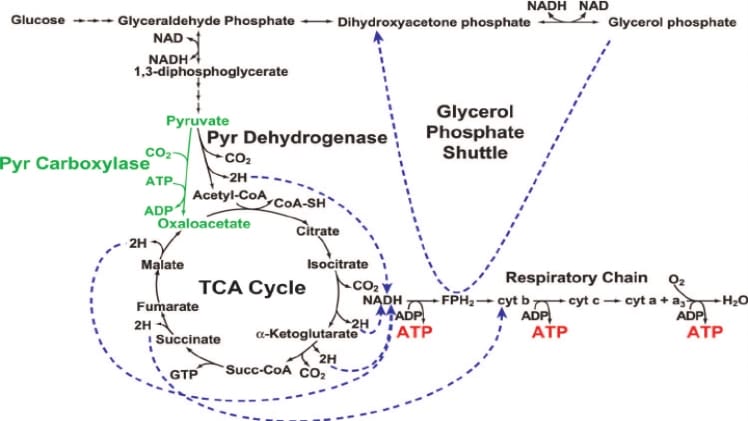
Pyruvate carboxylase (PC) is an enzyme that plays a critical role in the regulation of glycolysis and the metabolic pathways of both carbohydrates and fatty acids. It is responsible for catalyzing the carboxylation of pyruvate, a reaction that converts pyruvate into oxaloacetate, which is then used in the citric acid cycle. In order for this reaction to take place, PC must be activated. This article will discuss when PC is activated and the physiological conditions necessary for its activation.
Pyruvate Carboxylase Activation
PC is activated by adenosine triphosphate (ATP) and biotin. ATP binds to the enzyme, causing a conformational change that increases its affinity for biotin. Biotin then binds to the enzyme, allowing the carboxylation reaction to take place. In addition, PC is allosterically regulated by acetyl-CoA, which binds to the enzyme and increases its affinity for biotin.
Physiological Conditions for Activation
PC is activated in a variety of physiological conditions. In the presence of glucose, PC is activated in order to produce oxaloacetate, which is then used to produce acetyl-CoA that can be used in the citric acid cycle. In the presence of fatty acids, PC is activated in order to produce oxaloacetate, which is then used to produce citrate that can be used in the synthesis of fatty acids. PC is also activated during the process of gluconeogenesis, which is the synthesis of glucose from non-carbohydrate sources. Finally, PC is activated during the process of ketogenesis, which is the synthesis of ketone bodies from acetyl-CoA.
In summary, pyruvate carboxylase is an enzyme that catalyzes the carboxylation of pyruvate into oxaloacetate. It is activated by ATP and biotin, and is allosterically regulated by acetyl-CoA. PC is activated in a number of physiological conditions, including the presence of glucose, fatty acids, gluconeogenesis, and ketogenesis.