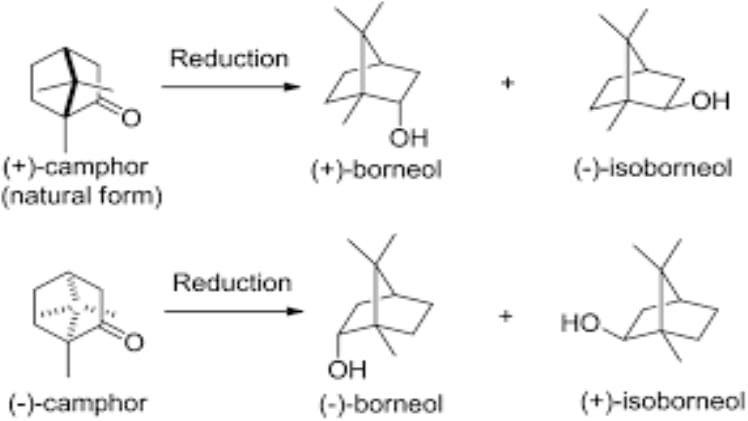
Borneol and Isoborneol are two closely related compounds that are often studied to gain insights into organic chemistry. These compounds display different properties, and it is important to understand how they compare in terms of stability. In this article, we will look at the differences between Borneol and Isoborneol, and investigate which one is more stable.
Comparing Borneol and Isoborneol
Borneol and Isoborneol are both monoterpenes, which are hydrocarbons consisting of two isoprene units. They are both cyclic molecules, containing a C10H18 skeleton. The main difference between the two compounds is in the arrangement of the isoprene units. Borneol has a cis arrangement of its isoprene units, while Isoborneol has a trans arrangement. This difference affects the shape of the molecule, and consequently, the way that the molecule interacts with other molecules.
Investigating Stability
The stability of a molecule is determined by its ability to resist change. In the case of Borneol and Isoborneol, the stability of the molecule is largely determined by its shape. Since Isoborneol has a more linear shape, it is more stable than Borneol, which has a more curved shape. This is because Isoborneol has fewer electrons in its pi system than Borneol, making it less prone to undergoing chemical reactions.
In addition, Isoborneol is more thermally stable than Borneol. This is because Isoborneol has a higher boiling point than Borneol. This is due to the fact that Isoborneol has stronger intermolecular forces, which require more energy to break.
Overall, Isoborneol is more stable than Borneol, both in terms of its shape and its thermal stability.
In conclusion, Isoborneol is more stable than Borneol. This is due to its more linear shape and stronger intermolecular forces. Understanding the stability of these compounds can be useful for designing new molecules with desired properties.