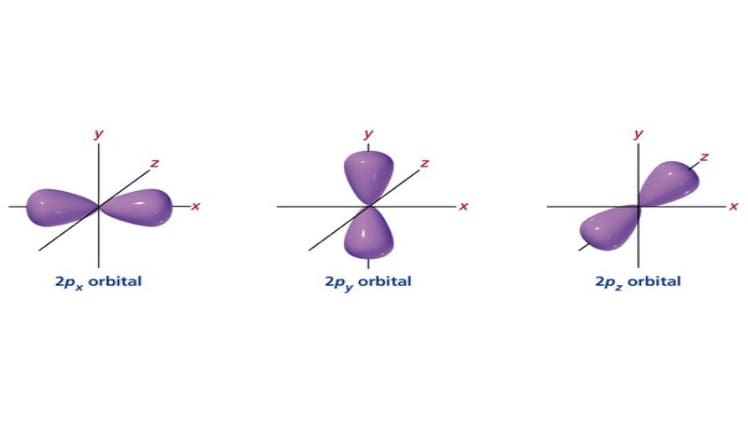
A 2d orbital is a mathematical concept that describes the probability of finding an electron in a two-dimensional space. This concept has been used in quantum mechanics to explain the behavior of electrons in atoms, molecules, and solids. In this article, we will discuss whether a 2d orbital is possible.
What is a 2d Orbital?
A 2d orbital is a mathematical representation of the probability of finding an electron in a two-dimensional space. This concept is used to describe the behavior of electrons in atoms, molecules, and solids. It is based on quantum mechanics, which states that electrons can exist in different energy levels and that the probability of finding an electron in a given energy level is given by the mathematical equation known as the Schrodinger equation.
The Schrodinger equation describes the behavior of an electron in a two-dimensional space by calculating the probability of finding the electron in a given energy level. This equation is used to calculate the probability of finding the electron in a given energy level. This equation is then used to calculate the probability of finding the electron in a given energy level in a two-dimensional space.
Is a 2d Orbital Possible?
The answer to this question is yes. A 2d orbital is possible in quantum mechanics. It is based on the Schrodinger equation, which describes the behavior of an electron in a two-dimensional space. The Schrodinger equation is used to calculate the probability of finding the electron in a given energy level. This equation is then used to calculate the probability of finding the electron in a given energy level in a two-dimensional space.
Furthermore, a 2d orbital is possible in the real world. For example, many molecules have a 2d orbital structure, which is used to describe the behavior of electrons in molecules. Additionally, certain materials such as graphene and nanotubes have a two-dimensional structure, which is based on a 2d orbital.
In conclusion, a 2d orbital is possible in quantum mechanics and the real world. It is based on the Schrodinger equation, which is used to calculate the probability of finding the electron in a given energy level in a two-dimensional space. This concept is used to describe the behavior of electrons in atoms, molecules, and solids.