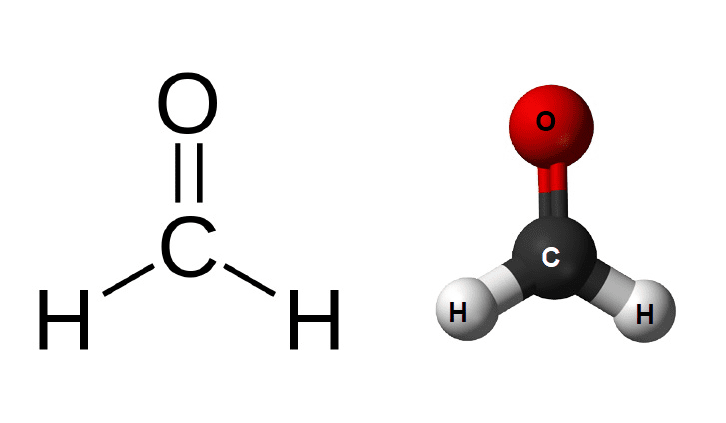
Formaldehyde is a colorless, flammable gas with a strong smell. It is widely used in manufacturing processes, as a preservative, and in household products. It is also a naturally occurring substance in the environment. Formalin and methanol are two compounds that are derived from formaldehyde, and they are often used interchangeably. In this article, we will explore the difference between formalin and methanol to better understand their similarities and differences.
What is Formaldehyde?
Formaldehyde is a colorless, flammable gas with a strong smell. It is a naturally occurring substance in the environment and is also widely used in manufacturing processes, as a preservative, and in household products. Formaldehyde is made up of carbon, hydrogen, and oxygen molecules and is also known as methanal, formalin, and methyl aldehyde. It has a wide range of uses, including in the production of plastics, adhesives, paints, fabrics, and paper products.
What is the Difference between Formalin and Methanol?
Formalin is a solution of formaldehyde in water, usually containing 10-40% formaldehyde by weight. It is used as a preservative and disinfectant, and is commonly used in laboratories and medical settings. Formalin is also used in embalming fluid and as a fixative in histology.
Methanol, also known as methyl alcohol, is a colorless liquid with a strong odor. It is a common ingredient in many industrial and consumer products, including paint strippers, antifreeze, and fuel. Methanol is also used in the production of formaldehyde and other chemicals.
Although both formalin and methanol are derived from formaldehyde, they are not synonymous. Formalin is a solution of formaldehyde in water, while methanol is a colorless liquid with a strong odor. Formalin is used as a preservative and disinfectant, while methanol is used in the production of formaldehyde and other chemicals.
In conclusion, formalin and methanol are not synonymous with formaldehyde. Formalin is a solution of formaldehyde in water, while methanol is a colorless liquid with a strong odor. Both compounds are derived from formaldehyde, but they have different uses. Understanding the difference between formalin and methanol is important in order to properly use them for their intended purposes.