Propene is a three-carbon alkene, also known as propylene, which is widely used in the production of plastics, synthetic rubber, and other materials. It is an important component of the petrochemical industry, and is also used as a fuel. In this article, we will look at the cis and trans isomers of propene and their implications.
Overview of Propene
Propene, or propylene, is a three-carbon alkene with the chemical formula C3H6. It is a colorless, odorless gas at room temperature. It is classified as a hydrocarbon because it is composed of only hydrogen and carbon atoms. It is an important component of the petrochemical industry, and is used in the production of plastics, synthetic rubber, and other materials. It is also used as a fuel.
Cis and Trans Isomers of Propene
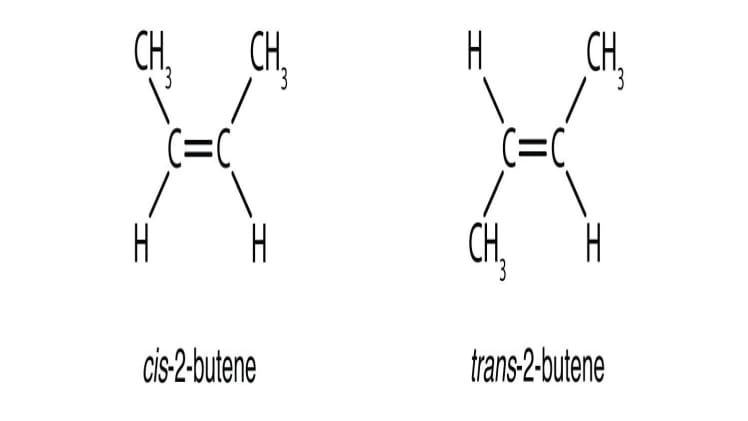
Propene can exist in two forms, known as cis and trans isomers. Cis isomers are molecules that have two identical groups of atoms on the same side of the double bond, while trans isomers have two identical groups of atoms on opposite sides of the double bond.
The two isomers of propene have different physical and chemical properties. The cis isomer has a boiling point of -9.8°C, while the trans isomer has a boiling point of -6.7°C. The cis isomer is also more soluble in water than the trans isomer. In terms of chemical reactivity, the cis isomer is more reactive than the trans isomer.
The two isomers of propene can also be interconverted. This process is known as isomerization, and is catalyzed by a variety of metals, such as aluminum, magnesium, and zinc. Isomerization is an important process in the petrochemical industry, as it allows the production of a variety of different products from the same starting material.
In conclusion, propene can exist in two forms, known as cis and trans isomers. These isomers have different physical and chemical properties, and can be interconverted through a process known as isomerization. Isomerization is an important process in the petrochemical industry, as it allows the production of a variety of different products from the same starting material.